 
Ī simple calculation reveals the limits of the numbers of water molecules that may be associated with an ion in a standard solution. 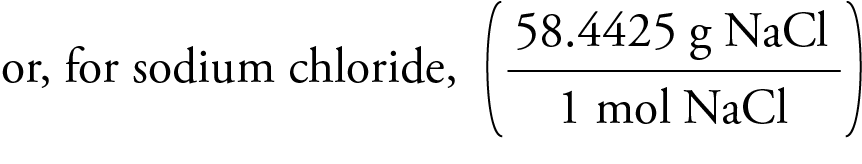
To do so, divide by the molar mass (which we discuss in Chapter 7). You must first convert the sodium chloride to moles. This problem gives you the value of your solute ( sodium chloride) in grams and the value of your solvent (water) in liters. 170 kD dextran is almost totally excluded while the lower molar mass polymers permeated the membrane to varying extents.įor sodium chloride ( relative atomic masses Na = 23.0, Cl = 35.5), the molar mass = 58.5 g mob1 therefore. Two molecular size dex-trans were used to probe the capsule permeability. The capsules were subsequently washed with PBS, coated for 15 min with 0.1% LV alginate (Kelco) and again washed in PBS. Capsules were made from 1.25% A-carrageenan (Fluka) and 0.02% carboxymethylcellulose (Aqualon) in 0.9% sodium chloride (core polymers) and 2% polydimethylamine-co-epichlorohydrin modified, quater-nized (Scientific Polymer Products) and a quaternary amine (Agefloc B50, CPS) in PBS (receiving bath) using a 3 min reaction time. Capsule permeability as measured by the inverse GPC method. Flence, if we measure out 58.44 g of sodium chloride, we obtain a sample that contains 1.000 mol NaCl formula units (Fig. E.2 was obtained in this way each sample contains the same number of atoms of the element (6.022 X 1023), but the masses vary because the masses of the atoms are different (Fig. It would be impossible to count out 6 X ID23 atoms of an element, but it is very easy to measure out a mass equal to the molar mass of the element in grams. Molar mass is important when we need to know the number of atoms in a sample. Alternatively about 2.9 g of the pure salt is accurately weighed out, dissolved in 500 mL of water in a graduated flask and the molar concentration calculated from the weight of sodium chloride employed. A 0.1000M solution is prepared by weighing out 2.922 g of the pure dry salt (see Section 10.74) and dissolving it in 500 mL of water in a graduated flask. Sodium chloride molar mass Sodium chloride has a relative molecular mass of 58.44.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
